High school chemistry | Unit 1 Start learning now! https://www.khanacademy.org/science/hs-chemistry/x2613d8165d88df5e:atoms-isotopes-and-ions
قد يعجبك أيضًا
Kids Songs by CoComelon
Primary 1 English
Alphabet (ABC) Songs by CoComelon
Learn Colors with CoComelon!
SS 1 English
Animation & Kids Songs collections For Babies | BabyBus
BabyBus | Kids Cartoon | Stories for Kids | #Reading
CoComelon & Friends Holiday and Christmas Videos for Kids | Little Baby Bum | Go Buster and More!
Year1 English
Baby Learning with CoComelon - Colors, Shapes and Numbers!
Let's learn about.... English Vocabulary for Kids
Primary 5 English
BEST SONGS for TODDLERS 👶🎵 (1 to 3 year olds)
BEST of CoComelon Bath Song + Wheels on the Bus
PrePrimary English
Masha & the Bear
Shapes, Colors, & Music by CoComelon
Year4 English
Family Fun by CoComelon
JJ & Friends by CoComelon
Baby Learning with CoComelon - Colors, Shapes and Numbers!
ABC Jamboree by StoryBots
CoComelon Lane | NEW Netflix Kids Show
Hank's Big Adventure!
التعليقات
8 تعليق
Courses on Khan Academy are always 100% free. Start practicing—and saving your progress—now! https://www.khanacademy.org/science/h... Want to explore more? Check out the full Atoms, isotopes, and ions playlist here: •Atoms,isotopes,andions|Chemistry|Kh... Lewis diagrams for atoms and ions are models that show the number of valence electrons (outermost electrons) they have. Lewis diagrams of atoms show the element's symbol surrounded by dots representing valence electrons. For monatomic ions, Lewis diagrams also includes brackets and the ion's charge. The number of valence electrons in an ion's Lewis diagram reflects the ion's charge. Sections: 00:00 - Introduction to Lewis diagrams 00:13 - Review of Bohr model using nitrogen 01:13 - Valence electrons and reactivity 01:34 - Drawing a Lewis diagram for nitrogen 02:07 - Lewis diagram for nitride ion 02:45 - Understanding periodic table group patterns 03:41 - Valence electrons and periodic table groups 04:03 - Shortcut for Groups
Courses on Khan Academy are always 100% free. Start practicing—and saving your progress—now! https://www.khanacademy.org/science/h... Want to explore more? Check out the full Atoms, isotopes, and ions playlist here: •Atoms,isotopes,andions|Chemistry|Kh... An atom with an electric charge is called an ion. The charge of an ion is equal to the number of protons minus the number of electrons. Sections: 00:00 - Question: Charge of calcium ion with 18 electrons 00:27 - Using atomic number to find protons 00:49 - Calculating calcium ion charge 01:22 - Neutral calcium vs. calcium ion 01:43 - Example: Ion with 7 protons and 10 electrons 02:16 - Identifying the ion as nitrogen 3− ------------------ Khan Academy is a nonprofit organization with the mission of providing a free, world-class education for anyone, anywhere. We offer quizzes, questions, instructional videos, and articles on a range of academic subjects, including math, biology, chemistry, physics, history, economics, fina
Courses on Khan Academy are always 100% free. Start practicing—and saving your progress—now: https://www.khanacademy.org/science/h... Want to explore more? Check out the full Atoms, isotopes, and ions playlist here: •Atoms,isotopes,andions|Chemistry|Kh... An atom with an electric charge is called an ion. A positively charged ion (a cation) has more protons than electrons. A negatively charged ion (an anion) has more electrons than protons. Sections: 00:00 - Introduction to ions 00:14 - Neutral atoms and charges 00:50 - Creating a carbon ion 01:50 - Example: Boron ion 02:46 - When atoms become ions 03:04 - Example question: Platinum isotope 03:45 - Calculating platinum ion charge 04:30 - Cations vs. anions 05:14 - Finding neutrons in platinum 06:07 - Summary: Atoms vs. ions ------------------ Khan Academy is a nonprofit organization with the mission of providing a free, world-class education for anyone, anywhere. We offer quizzes, questions, instructional videos, and article
Courses on Khan Academy are always 100% free. Start practicing—and saving your progress—now! https://www.khanacademy.org/science/a... Want to explore more? Check out the full Atoms, isotopes, and ions playlist here: •Atoms,isotopes,andions|Chemistry|Kh... The average atomic mass (sometimes called atomic weight) of an element is the weighted average mass of the atoms in a naturally occurring sample of the element. Average masses are generally expressed in unified atomic mass units (u), where 1 u is equal to exactly one-twelfth the mass of a neutral atom of carbon-12. Sections: 00:15 - Understanding the atomic scale 00:48 - Measuring mass with atomic mass units 01:36 - Definition of unified atomic mass unit 02:39 - Mass of protons, neutrons, and electrons 03:31 - Atomic mass and the periodic table 04:27 - Isotopes of hydrogen 05:45 - Atomic mass as a weighted average 07:10 - Average atomic mass vs. atomic weight 07:56 - Relative atomic mass explained ------------------ Khan Ac
Courses on Khan Academy are always 100% free. Start practicing—and saving your progress—now! https://www.khanacademy.org/science/h... Want to explore more? Check out the full Atoms, isotopes, and ions playlist here: •Atoms,isotopes,andions|Chemistry|Kh... The sum of the number of neutrons and protons in an atom is called its mass number. If we know the mass number of an atom, we can determine the number of neutrons in its nucleus. Sections: 00:17 - Finding neutrons in a sodium isotope 00:40 - Rearranging the mass number equation 00:58 - Solving for neutrons in sodium-23 01:11 - Example: Neutrons in platinum-195 01:43 - Solving for neutrons in Pt-195 ------------------ Khan Academy is a nonprofit organization with the mission of providing a free, world-class education for anyone, anywhere. We offer quizzes, questions, instructional videos, and articles on a range of academic subjects, including math, biology, chemistry, physics, history, economics, finance, grammar, preschool
Courses on Khan Academy are always 100% free. Start practicing—and saving your progress—now! https://www.khanacademy.org/science/h... Want to explore more? Check out the full Atoms, isotopes, and ions playlist here: •Atoms,isotopes,andions|Chemistry|Kh... Isotopes are atoms of the same element with different numbers of neutrons. Since isotopes of an element have different numbers of neutrons, they also have different masses. The sum of the number of neutrons and protons in an atom is called its mass number. Isotopes can be represented in writing using isotope notation. Sections: 00:00 - Elements and atomic number 00:29 - What are isotopes? 00:59 - Introduction to isotope notation 01:24 - Isotope notation for carbon 02:49 - Hyphen notation for isotopes 03:26 - Recap: Isotopes and their notations Khan Academy is a nonprofit organization with the mission of providing a free, world-class education for anyone, anywhere. We offer quizzes, questions, instructional videos, and articl
Courses on Khan Academy are always 100% free. Start practicing—and saving your progress—now! https://www.khanacademy.org/science/h... Want to explore more? Check out the full Atoms, isotopes, and ions playlist here: •Atoms,isotopes,andions|Chemistry|Kh... Atoms come in a variety of types, called elements, each with a unique set of physical and chemical properties. An element is defined by the number of protons in its atoms, known as its atomic number. The periodic table organizes the elements by atomic number and provides other key information about them such as their names and chemical symbols. Sections: 00:00 - What are elements? 00:32 - Atomic number and protons 01:03 - Electrons in neutral atoms 01:33 - Introduction to the periodic table 01:59 - Chemical symbols of elements 02:45 - Understanding an element's box 03:34 - Using the periodic table 03:36 - Example: Phosphorus 03:55 - Example: Mercury 04:16 - Conclusion: Elements in the universe ------------------ Khan Academ
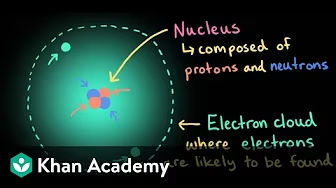
Courses on Khan Academy are always 100% free. Start practicing—and saving your progress—now! https://www.khanacademy.org/science/h... Want to explore more? Check out the full Atoms, isotopes, and ions playlist here: •Atoms,isotopes,andions|Chemistry|Kh... Atoms are made up of three types of subatomic particles: protons, neutrons, and electrons. Protons and neutrons are found in the nucleus, the dense region at the center of an atom. Electrons are found outside the nucleus. Protons are positively charged and have a mass of about 1 u. Neutrons are neutral (have no charge) and also have a mass of about 1 u. Electrons are negatively charged and have a much smaller mass of about 0.0005 u. Sections: 00:00 - Introduction to atoms 00:19 - Atoms as building blocks of matter 00:40 - Structure of the atom 01:08 - Charges of subatomic particles 01:39 - Masses of subatomic particles 02:12 - Atoms make up everything 02:25 - Summary: Subatomic particles in all atoms ------------------ Khan